On this page
Healthcare Provider Guidance and Resources
- CDC Infection Control Recommendations for Healthcare Professionals During COVID-19
- NIH Clinical Guidelines: Therapies for High-Risk, Nonhospitalized Patients
- NIH Panel's Statement on Prioritizing the Use of Outpatient Therapies
- CDC Guidance for Healthcare Personnel with Potential COVID-19 Exposure
- Handout: What to Do After You Have Been Tested for COVID-19
- Handout: Home Isolation for Individuals with COVID-19
- CDC guidance related to COVID-19
Patient Handouts
Vaccination Providers
Reporting COVID-19
- Updated SARS-CoV-2 Reporting Requirements May 25, 2023
Health Facilities
Specimen Collection, Delivery and Reporting
- COVID-19 Specimen Collection
- COVID-19 Lab Requisition Form
- COVID-19 Refrigerated Specimen Packaging
- COVID-19 Frozen Specimen Packaging
- COVID-19 Specimen Transport and Delivery
Specimen Reporting
SARS-CoV-2 Virus Strain Surveillance
- COVID-19 Enhanced Surveillance Submission Recommendations
- SARS-CoV-2 Enhanced Surveillance Submission Requisition Form 1198
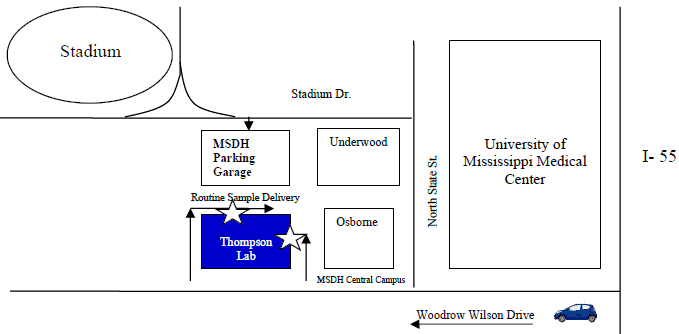
Map and Directions for Laboratory Dropoff
The Dr. F.E.”ED” Thompson, Jr. Public Health Laboratory Facility is on the State Department of Health's central office campus in Jackson, MS at 350 East Woodrow Wilson Drive, Jackson, 39216.
Local Delivery of Samples: Drop samples off at the rear of the Thompson lab building. Detailed instructions are in our sample shipping guidance.
Dropoff hours: Monday through Friday from 8:00–4:30.
CDC Guidance
Healthcare and EMS
- Home Care Guidance
- Discontinuation of Home Isolation for Persons with COVID-19
- Healthcare Infection Control Guidance
- Clinical Care Guidance
- Healthcare Personnel with Potential COVID-19 Exposure Guidance
- Inpatient Obstetric Healthcare Guidance
- Guidance for EMS
Contact Tracing
- Contact Tracing Information and Guidance
- Collection and Submission of Postmortem Specimens from Deceased Persons with Confirmed or Suspected COVID-19